Basics of Fluorescence Lifetime Imaging and FLIM Applications to FRET Biosensors

Dr. Luis Alvarez
Read BioDr. Luis Alvarez studied physical chemistry at the Université Paris-Sud XI in Orsay, where he worked on molecular quantitative imaging in live cells. He concentrated on fluorescence lifetime imaging and the effects of reactive oxygen species (ROS) on fluorescent proteins. In 2010, Luis moved to Dublin, where he worked on host-pathogen interactions at University College Dublin and the National Children Research Centre and studied how mucosal immunity uses ROS to respond to bacterial pathogen infections. During this time, he developed 3D infection models from ex-vivo organ cultures and organoids. In 2016, Luis moved to the University of Oxford, where he developed advanced quantitative imaging approaches to study host-pathogen interactions. This work centered on understanding HIV and EBOLA entry mechanisms into cells. Luis joined Leica Microsystems in 2019 as a product application manager for functional imaging.
CloseIn this tutorial you will learn:
- How researchers (Anzilotti 2019) used FLIM to elucidate the mechanisms responsible for a B cell deficiency
- A new approach to measure the cellular function of the Zn2+ transporter ZIP7 in primary mouse cells, comparing wild-type and CRISPR-Cas9 derived mutant cells with the help of the eCALWY-4 and eCALWY-6 biosensors (Hessels 2016).
- How the Leica SP8 FALCON FLIM setup allowed to properly measure the Zn2+homeostasis in these cells.
Tutorial abstract
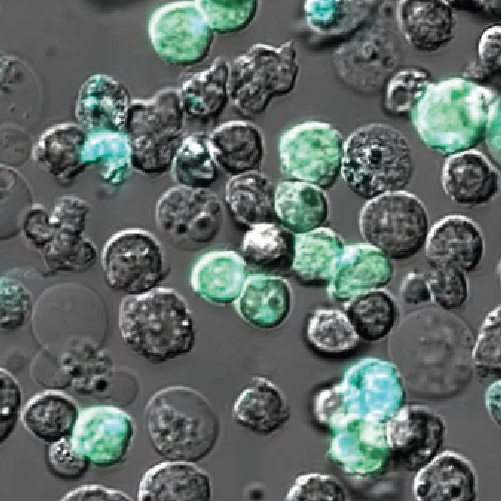
Functional imaging is a rapidly growing field key to driving new understanding in biology. Insights into the function and interaction of molecules are the key to reveal the underlying cellular mechanisms. In this context, fluorescence lifetime imaging (FLIM) is a powerful tool, providing valuable information beyond spectral imaging. FLIM is immune to concentration artefacts and sensitive to molecular environment such has pH changes, ion concentrations, and more.
Förster Resonance Energy Transfer (FRET) is an example of molecular environmental changes. The donor lifetime is shortened by the presence of the acceptor. FRET experiments thus benefit from FLIM information. The FRET-FLIM readout is independent of the donor or acceptor concentration making it the quantification assay of choice. Recently, FRET has been exploited to engineer sensors (FRET biosensors). FRET signal changes in these biosensors correspond to either binding or release of a ligand. How this type of readout can help uncover biological mechanisms was recently shown in a Nature Immunology article (Anzilotti 2019). The approach used in this article, and described in this webinar, opens the field to imaging in a range of situations in primary cells, where sensitivity and the need to avoid damaging the cells are both paramount.
References
Anzilotti et. al An essential role for the Zn2+ transporter ZIP7 in B cell development. Accepted manuscript Nature Immunology 2019 DOI. 10.1038/s41590-018-0295-8
Hessels AM, Taylor KM, Merkx M. Monitoring cytosolic and ER Zn(2+) in stimulated breast cancer cells using genetically encode FRET sensors. Metallomics. 2016 Feb;8(2):211-7. doi: 10.1039/c5mt00257e.